Looking through my collection I was dazzeled by a 7 carat yellow gem. This gem sparkles more than my wifes diamond. I checked the refractive index but was beyond my machine over 1.8. My themal capacity meter read garnet. So does anyone think this this could be a Topazolite garnet.
Thanks
With the larger carat size and the high luster this stone has i do not think it is a garnet. I am more inclined to believe that it is a yellow zircon.
Thanks will have to do further tests
Hi looked through a high power Lieca zoom mike but no doubling seen in the cullets tried through the side facets again could not see any. I am away from my digital spectroscope and my handheld so will have to do more tests without them.
Very interesting
Hi there dear friend. So just from the picture! I would say that it is a lab grown gem don’t get me wrong it could still be something, but if I had to guess it could be a topazolite or sphalerite. As I have said a few times in different responses or this amazing forum you can’t really tell just by picture but, I get a pretty good idea every now and then and do my best to answer as gently and as honestly as possible every time. I do hope that it is a topazolite for your sake but do keep us updated with your findings after firther testing. Best regards Jarryed.
Hi did some spectroscopy but still not sure. I have better instruments at home and that may help
Thanks
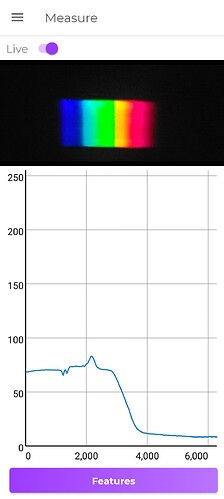
First picture is yellow gem
Second is CFL bulb for calibration
I am pretty sure it’s a zircon. Pretty though
the spectra reminds me of the old “depression glass”… Pb-enriched. Check the R.I. leaded-glass is usually around 2.15… zircon is about 1.95 … and garnet should be less than 1.80.
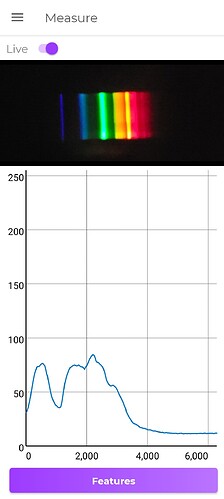
New spectra from a different spec
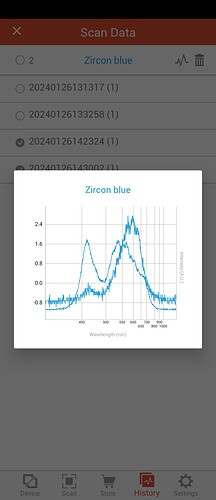
First shows yellow gem and a blue zircon (probably phony)
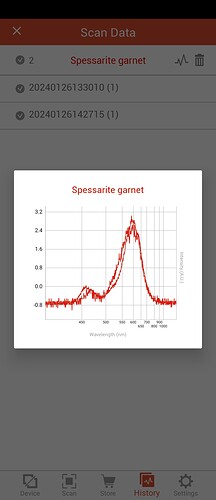
Second shows yellow gem and spessarite garnet (don’t have Andradite garnet)
refractive index for garnet is 1.73 to 1.759 for Zircon it’s 1.93 to 1.98. Zircon is birefringent while garnet is not. I would vote for zircon with the others.
@StevenH26783 it was my understanding that some varieties of garnet can read above index (1.80)?
Some garnets can have RIs over 1.80. This includes garnets of the andradite variety which would include topazolite. This article contains a table that compares the RIs, colors, and spectra of some garnet species and blends.
thanks for the professional information… am I correcting in saying that andradite, grossular and uvaroite belong the ugrandite family of calcium garnets with Al, Fe, and Cr substitution for each other. Andradite would be the Fe3+ end member grossular being the Al+3 end member. None of them occur as a pure endmembers. I also have read that there’s some 2+ My question is how close to the andradite endmember is topazolite? some scholarly sources show limited divalent ion substitution with the pyralspite group. Mg+2 has an ionic radius that is too small. The transition metals following Ca+2 have similar sized radii…but ae not in the divalent site…I would also presume that a high Fe content would give a garnet a higher RI and SpG. Am I correct or not?.. or it it more complicated than that?.
Hi again I have picture of the yellow gem above a thin pencil line. Sorry for the poor quality I am having trouble with a new digital mic
whatever you have is an attractive stone. Could be quite valuable if it can be definitively ID’s as topazolite. Good luck
Just from the look, I would guess Sphene.
Hi thanks for the response, even if it is a flawless 7 carat sphene I would be very happy. It has been kicking around my collection loose for a long time with no visible damage. I am snowbirding at present but I will check the stone with my very sensitive Geiger counter when I return home.
Thanks again
unless there’s a lot of thorium substituting for calcium in titanite, it might not be highly radioactive. titianite is mildly radioactive at best. what is the specific gravity?
I would look for doubling, if it doubles, it is most likely zircon.
Have you considered sphene (titanite)?